The recent approval by the Food and Drug Administration of Moderna’s vaccine for respiratory syncytial virus (RSV) for adults aged 60 and above marks a significant milestone in the company’s journey to diversify its product offerings. This approval comes at a crucial time for Moderna, as it seeks to address the declining demand for its Covid vaccine, which is currently its only commercially available product. The decision to approve Moderna’s RSV vaccine is based on a late-stage trial involving older adults, who are particularly susceptible to severe cases of RSV.
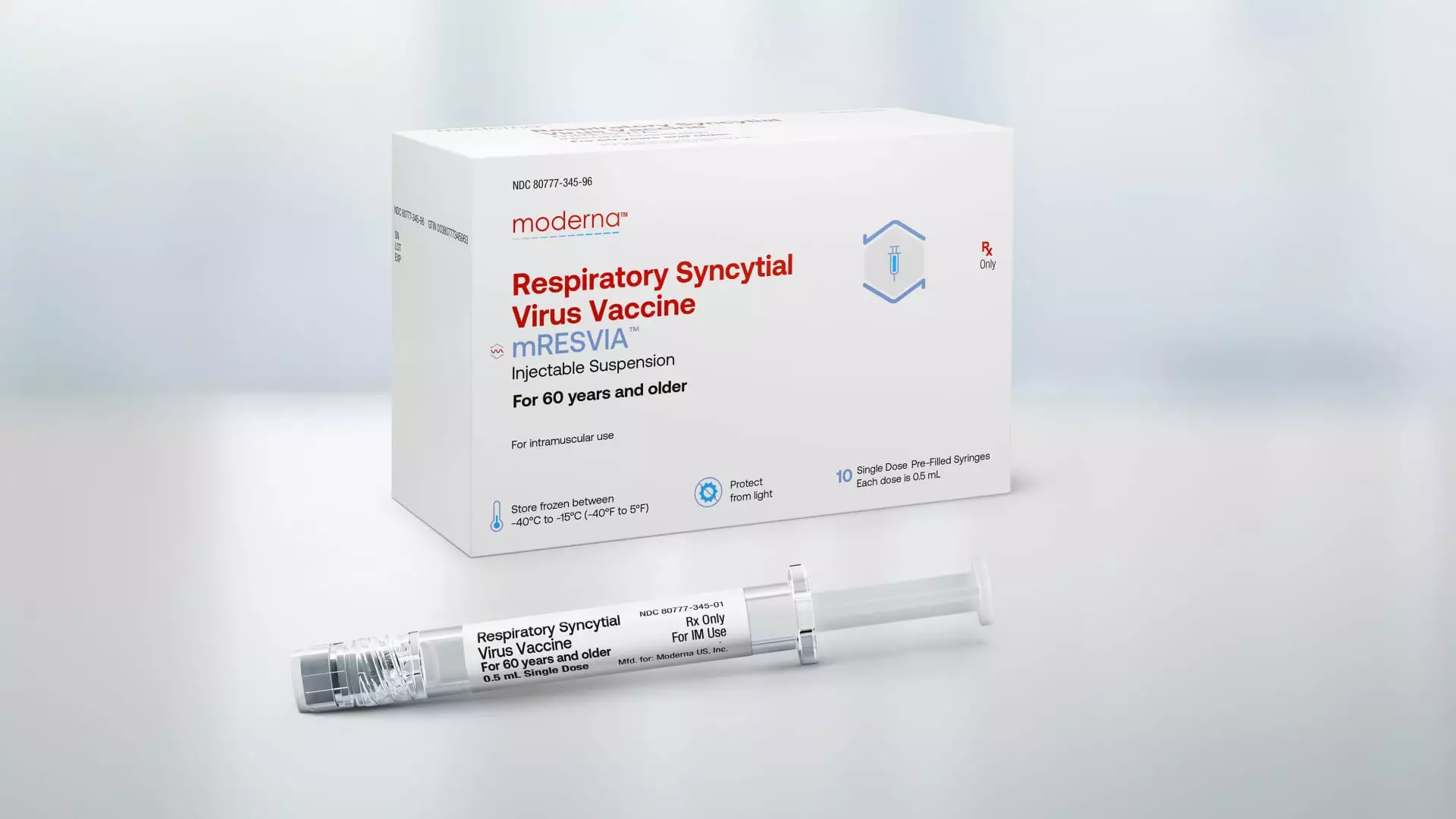
Moderna’s RSV vaccine, marketed under the brand name mRESVIA, is the first messenger RNA vaccine to receive FDA approval for a disease other than Covid. What sets Moderna’s vaccine apart is that it is the only RSV vaccine available in a pre-filled syringe, making it easier to administer to patients. This unique feature could potentially give Moderna a competitive edge against existing RSV vaccines from pharmaceutical giants like GSK and Pfizer.
The approval of Moderna’s RSV vaccine underscores the versatility of the company’s messenger RNA platform beyond just treating Covid. Moderna is leveraging this groundbreaking technology to develop vaccines for a range of diseases, including cancer, norovirus, and now RSV. With over 40 products in development and several in late-stage trials, Moderna is positioning itself as a leader in the biotech industry.
Despite facing setbacks in 2023, Moderna’s stock has rebounded significantly in 2024, with shares soaring over 40% this year. Investors are optimistic about the long-term potential of Moderna’s mRNA product pipeline, particularly with the successful approval of the RSV vaccine for older adults. Moderna’s projected full-year sales in 2024 are estimated to reach around $4 billion, with revenue from the RSV vaccine contributing to this growth.
Challenges and Opportunities Ahead
While Moderna’s RSV vaccine has shown promising efficacy in preventing symptoms of the virus, concerns have been raised about the decline in effectiveness over time compared to other vaccines on the market. Moderna has emphasized that direct comparisons are challenging due to differences in study populations, geographic locations, and case definitions for RSV. Moving forward, Moderna is focused on expanding its product portfolio to include a combination shot targeting Covid and the flu, as well as a personalized cancer vaccine in collaboration with Merck.
The FDA approval of Moderna’s RSV vaccine represents a significant breakthrough in the fight against infectious diseases, particularly in older adults who are at a higher risk of severe RSV-related complications. Moderna’s innovative use of mRNA technology opens up new possibilities in vaccine development and positions the company for continued growth and success in the years to come. With a strong focus on research and development, Moderna is poised to make a lasting impact on public health and shape the future of healthcare.

Leave a Reply